Introduction
Pollution aspects related to the ceramic industry are mainly due to dust emission both in workplace and in ambient air. Effluents are characterized by higher concentration of suspended solids.
Large volumes of wastewater are generated during the process. Process wastewater is generated mainly when clay materials are flushed out and suspended in flowing water during the manufacturing process and equipment cleaning. The added directly to ceramic body mixes; is subsequently evaporated into the air during the drying and firing stages.
The pollutants in the Ceramic wastewater are different suspended solids, dissolved solids, chemical oxygen demand and Low Biochemical oxygen demand. This wastewater caused serious environmental problems due to their high color, large amount of suspended solids, and high chemical oxygen demand. So, they have to be removed before being discharged into the environment.
Limits of pollutants in wastewater are depending on the type of receiving water body. The parameters that should be monitored and/or inspected are biochemical oxygen demand BOD, chemical oxygen demand COD, pH, temperature, total suspended solids TSS, and total dissolved solids TDS,
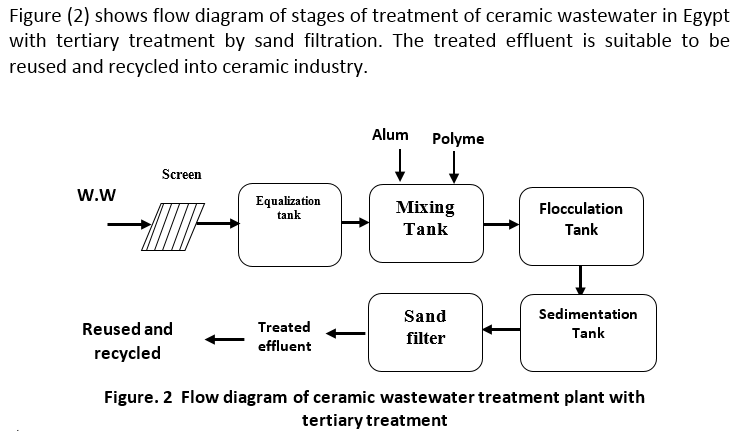
The problem of ceramic wastewater utilization may be solved with different treatment techniques for reuse or recycle / treated wastewater and recovery of treated water.
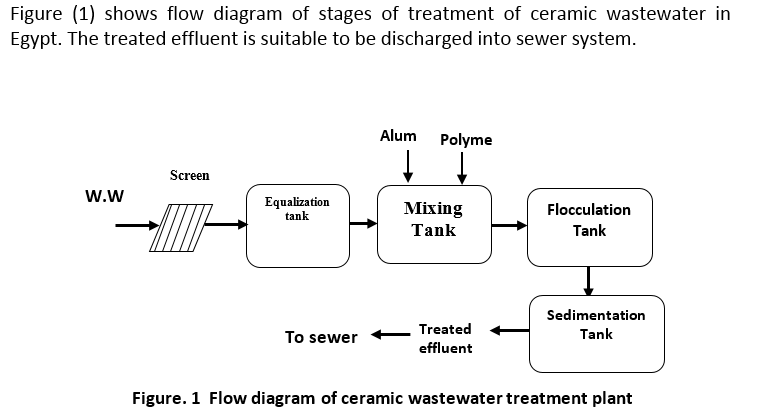
Different physicochemical treatments have been used to treat the Ceramic wastewater such as coagulation with mineral coagulants and organic coagulants aids followed by sedimentation and sand filtration. The proposed system of the study under investigation is to treat the effluent for reuse or discharge into the receiving body needs the following units: screens, equalization tank, and feed pump, flow regulator, mixing tank, flocculation tank and clarifier.
Effluents of Ceramic Industry
Effluents of ceramic industry are characterized by:
- Highest levels of water pollution that generated from washing of molds and final products to remove any suspended impurities on the pieces. Impurities are removed after the pouring stage and before the drying stage.
- Spent lube oil from garage and workshops if discharged to sewer will give oily wastewater.
- Tile polishing generates a large quantity of wastewater high in suspended solids and settable solid.
- Industrial wastewater.
Typical effluent characteristics of the Egyptian ceramic industry are shown in the following Table 1
Table1: Typical effluent characteristics of the Egyptian ceramic industry [1].
| Parameter | Average analysis (mg/liter) |
| pH | 7.5 |
| Total suspended solid | 700 |
| Total dissolved solid | 220 |
| Biological oxygen demand | 30 |
| Chemical oxygen demand | 400 |
| Oil and grease | 25 |
Effluent guidelines are applicable for direct discharges of treated effluents to surface waters for general use. Site-specific discharge levels may be established based on the availability and conditions in the use of publicly operated sewage collection and treatment systems or, if discharged directly to surface waters.
Method of Treatment of Industrial Wastewater of Ceramic Industry
Treatment of industrial wastewater of resulting from the ceramic manufacturing process mainly depends on physical and chemical treatment.
PHYSICAL TREATMENT PROCESSES
Among the first treatment methods used were physical unit operations, in which physical forces are applied to remove contaminates. Today they still form the basis of most process flow systems for wastewater treatment. This section briefly discusses the most commonly used physical unit operations.
- Screening
The screening of wastewater, one of the oldest treatment methods, removes gross pollutants from the waste stream to protect downstream equipment from damage, avoid interference with plant operations and prevent objectionable floating material from entering the primary settling tanks. Screening devices may consist of parallel bars, rods or wires, grating, wire mesh, or perforated plates, to intercept large floating or suspended material. The openings may be of any shape, but are generally circular or rectangular. The material retained from the manual or mechanical cleaning of bar racks and screens is referred to as “screenings”, and is either disposed of by burial or incineration, or returned into the waste flow after grinding [2].
- Flow Equalization
Flow equalization is a technique used to improve the effectiveness of secondary and advanced wastewater treatment processes by leveling out operation parameters such as flow, pollutant levels and temperature over a period of time. Variations are damped until a near-constant flow rate is achieved, minimizing the downstream effects of these parameters. Flow equalization may be applied at a number of locations within a waste-water treatment plant, e.g. near the head end of the treatment works, prior to discharge into a water body, and prior to advanced waste treatment operations [2].
- Sedimentation
Sedimentation, a fundamental and widely used unit operation in waste-water treatment, involves the gravitational settling of heavy particles suspended in a mixture. This process is used for the removal of grit, particulate matter in the primary settling basin, biological floc in the activated sludge settling basin, and chemical flow when the chemical coagulation process is used. Sedimentation takes place in a settling tank, also referred to as a clarifier. There are three main designs, namely, horizontal flow, solids contact and inclined surface. In designing a sedimentation basin, it is important to bear in mind that the system must produce both a clarified effluent and a concentrated sludge. Four types of settling occur, depending on particle concentration: discrete, flocculent, and hindered and compression. It is common for more than one type of settling to occur during a sedimentation operation [2].
- Sand filtration
The filtration of effluents from waste-water treatment processes is a relatively recent practice, but has come to be widely used for the supplemental removal of suspended solids from waste-water effluents of biological and chemical treatment processes, in addition to the removal of chemically precipitated phosphorus. The complete filtration operation comprises two phases: filtration and cleaning or backwashing. The waste-water to be filtered is passed through a filter bed consisting of granular material (sand, anthracite and/or garnet), with or without added chemicals. Within the filter bed, suspended solids contained in the waste-water are removed by means of a complex process involving one or more removal mechanisms such as straining, interception, impaction, sedimentation, flocculation and adsorption. The phenomena that occur during the filtration phase are basically the same for all types of filters used for waste-water filtration. The cleaning/backwashing phase differs, depending on whether the filter operation is continuous or semi continuous. In semi-continuous filtration, the filtering and cleaning operations occur sequentially, whereas in continuous filtration the filtering and cleaning operations occur simultaneously [3].
CHEMICAL TREATMENT PROCESSES
Chemical processes used in wastewater treatment are designed to bring about some form of change by means of chemical reactions. They are always used in conjunction with physical unit operations and biological processes. In general, chemical unit processes have an inherent disadvantage compared to physical operations in that they are additive processes. That is to say, there is usually a net increase in the dissolved constituents of the wastewater. This can be a significant factor if the wastewater is to be reused. This section discusses the main chemical unit processes, including chemical precipitation, adsorption, disinfection, dechlorination and other applications.
- Chemical precipitation
Chemical coagulation of raw waste-water before sedimentation promotes the flocculation of finely divided solids into more readily settleable flocs, thereby enhancing the efficiency of suspended solid, BOD5 and phosphorus removal as compared to plain sedimentation without coagulation. The degree of clarification obtained depends on the quantity of chemicals used and the care with which the process is controlled [4].
Coagulant selection for enhanced sedimentation is based on performance, reliability and cost. Performance evaluation uses jar tests of the actual waste-water to determine dosages and effectiveness. Chemical coagulants that are commonly used in waste-water treatment include alum (Al2(SO4)3.14.3H2O), ferric chloride (FeCl3.6H2O), ferric sulfate (Fe2(SO4)3), ferrous sulfate (FeSO4.7H2O) and lime (Ca(OH)2). Organic polyelectrolyte is sometimes used as flocculation aids [5].
Suspended solids removal through chemical treatment involves a series of three unit operations: rapid mixing, flocculation and settling. First, the chemical is added and completely dispersed throughout the wastewater by rapid mixing for 20-30 seconds in a basin with a turbine mixer. Coagulated particles are then brought together via flocculation by mechanically inducing velocity gradients within the liquid. Flocculation takes 15 to 30 minutes in a basin containing turbine or paddle-type mixers.
The advantages of coagulation include greater removal efficiency, the feasibility of using higher overflow rates, and more consistent performance. On the other hand, coagulation results in a larger mass of primary sludge that is often more difficult to thicken and dewater. It also entails higher operational costs and demands greater attention on the part of the operator.
- Disinfection
Disinfection refers to the selective destruction of disease-causing micro-organisms. This process is of importance in wastewater treatment owing to the nature of wastewater, which harbors a number of human enteric organisms that are associated with various waterborne diseases. Commonly used means of disinfection include the following:
(i) Physical agents such as heat and light;
(ii) Mechanical means such as screening, sedimentation, filtration, and so on;
(iii) Radiation, mainly gamma rays;
(iv) Chemical agents including chlorine and its compounds, bromine, iodine, ozone, phenol and phenolic compounds, alcohols, heavy metals, dyes, soaps and synthetic detergents, quaternary ammonium compounds, hydrogen peroxide, and various alkalis and acids. The most common chemical disinfectants are the oxidizing chemicals, and of these, chlorine is the most widely used.
Disinfectants act through one or more of a number of mechanisms, including damaging the cell wall, altering cell permeability, altering the colloidal nature of the protoplasm and inhibiting enzyme activity. In applying disinfecting agents, several factors need to be considered: contact time, concentration and type of chemical agent, intensity and nature of physical agent, temperature, number of organisms, and nature of suspending liquid [5].
By
Ahmed Ahmed Elserwy
Water & Environmental Consultant
Ain Shames University, Faculty of Science.
References
[1] Egyptian Environmental Affairs Agency (EEAA); Egyptian Pollution Abatement Project (EPAP) Inspection Manual of Ceramic Industry, 2003.
[2] Economic and Social Commission for Western Asia, “Wastewater Treatment Technologies a General Review “, United Nation, 11 September 2003.
[3] Metcalf and Eddy, Inc. Wastewater Engineering: Treatment Disposal and Reuse, third edition. New York: McGraw-Hill, 1991.
[4] Metcalf and Eddy, Inc. Wastewater Engineering: Treatment and Reuse, fourth edition. New York: McGraw-Hill, 2003.
[5] Qasim, S.R. , Wastewater Treatment Plants: Planning, Design, and Operation (Lancaster, Pennsylvania: Technomic Publishing Company, 1999).