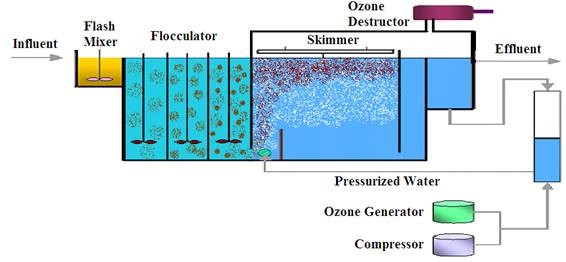
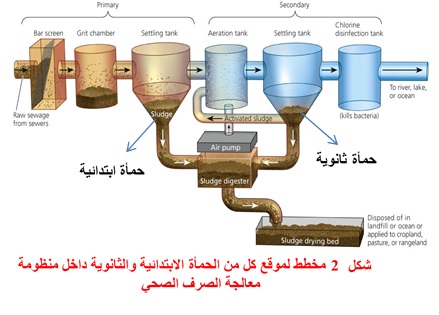
1- Introduction
New water-treatment goals for disinfection by-products (DBP) and for microbial inactivation have increased the need for new disinfection technologies. Water and wastewater systems will need to use disinfection methods that are effective for killing pathogens without forming excessive DBP [1]. Ozone is an attractive alternative. This technology has evolved and improved in recent years, thereby increasing its potential for successful application. In August 1997, the US Environmental Protection Agency (US EPA) listed ozone as a “compliance” in the requirements of the Surface Water Treatment Rule for all three sizes of drinking water systems [2]. Many of the existing facilities using chlorination may be required to upgrade their present disinfection systems to install alternative disinfection processes.
Ozone (O3) is a molecule that can co-exist with air or high-purity oxygen, or can dissolve in water. It is a very strong oxidizing agent and a very effective disinfectant.
Ozone is a colorless gas that has an odor most often described as the smell of air after a spring electrical thunderstorm. Some people also refer to the odor as similar to the smell of watermelons. Actually, ozone owes its name to its odor. The word ozone is derived from the Greek word ozein [3]. Ozone is an extremely unstable gas. Consequently, it must be manufactured and used onsite. It is the strongest oxidant of the common oxidizing agents. Ozone is manufactured by passing air or oxygen through two electrodes with high, alternating potential difference.
Ozone is a very strong oxidizing agent, having an oxidation potential of 2.07 V [4].
Ozone will react with many organic and inorganic compounds in water or wastewater.
These reactions are typically called “ozone demand” reactions. They are important in ozone disinfection system design because the reacted ozone is no longer available for disinfection. Waters or wastewaters that have high concentrations of organics or inorganics may require high ozone dosages to achieve disinfection. It is very important to conduct pilot plant studies on these wastewaters during ozone disinfection system design in order to determine the ozone reaction kinetics for the level of treatment prior to ozone disinfection.
- Reactions of Ozone with Organic Compounds
Wastewater is composed mainly by organic compounds which are biodegradable and non-biodegradable. Several investigators developed an in-depth analysis of the reactions for ozone with various organic compounds. These reactions were described by Miller et al. [4], and are summarized below:
(a) Aromatic compounds: Phenol reacts readily with ozone in aqueous solution. Oxalic and acetic acids are relatively stable to ozonation in the absence of a catalyst such as ultraviolet light or hydrogen peroxide. Cresols and xylenols undergo oxidation with ozone at faster rates than does phenol. Pyrene, phenanthrene, and naphthalene oxidize by ring rupture.
Chlorobenzene reacts with ozone slower than does phenol.
(b) Aliphatic compounds: There is no evidence that ozone reacts with saturated aliphatic hydrocarbons under water- or wastewater-treatment conditions. There is no evidence that ozone oxidizes trihalomethanes. Ozone combined with ultraviolet radiation does oxidize chloroform to produce chloride ion, but no identified organic oxidation product. Unsaturated aliphatic or alicyclic compounds react with ozone.
(c) Pesticides: Ozonation of parathion and malathion produces paraoxon and malaoxon,respectively, as intermediates, which are more toxic than are the starting materials.
Continued ozonation degrades the oxons, but requires more ozone than the initial reaction.
Ozonation of heptachlor produces a stable product not yet identified. Aldrin and 2,4,5-T are readily oxidized by ozone, but dieldrin, chlordane, lindane, DDT, and endosulfan are only slightly affected by ozone.
(d) Humic acids: Humic materials are resistant to ozonation, requiring long ozonation times to produce small amounts of acetic, oxalic, formic, and terephthalic acids, carbon dioxide, and phenolic compounds. Ozonation of humic materials followed by immediate chlorination (within 8 min) has been shown to reduce trihalomethane formation in some cases. Ozonized organic materials generally are more biodegradable than the starting, unoxidized compounds.
- Advantages and Disadvantages
It is important to note that ozone, like other technologies, has its own set of advantages and disadvantages that show up in differing degrees from one location to the next [5].
Using ozone has the following advantages:
(a) Possesses strong oxidizing power and requires short reaction time, which enables the pathogens to be killed within a few seconds.
(b) Produces no taste or odor.
(c) Provides oxygen to the water after disinfecting.
(d) Requires no chemicals.
(e) Oxidizes iron and manganese.
(f ) Destroys and removes algae.
(g) Reacts with and removes all organic matter.
(h) Decays rapidly in water, avoiding any undesirable residual effects.
(i) Removes color, taste, and odor producing compounds.
(j) Aids coagulation by destabilization of certain types of turbidity.
Among the disadvantages of using ozone are the following:
(a) Toxic (toxicity is proportional to concentration and exposure time).
(b) Cost of ozonation is high compared with chlorination.
(c) Installation can be complicated.
(d) Ozone-destroying device is needed at the exhaust of the ozone reactor to prevent toxicity.
(e) May produce undesirable aldehydes and ketones by reacting with certain organics.
(f ) No residual effect is present in the distribution system, thus postchlorination may be required.
(g) Much less soluble in water than chlorine; thus, special mixing devices are necessary.
(h) It will not oxidize some refractory organics or will oxidize too slowly to be of practical
significance.
- APPLICATIONS OF OZONE
Ozone acts both as a very strong oxidizing agent and as a very effective disinfectant.
Consequently, it has multiple uses in potable water treatment, wastewater renovation, cooling water towers, groundwater remediation, and industrial waste treatment. The
following is a snapshot description of each of its applications (5–8).
4.1. Disinfection against Pathogens
Transfer of ozone into the water is the first step in meeting the disinfection objective, because ozone must be transferred and residual oxidants produced before effective disinfection will occur [9]. Once transferred, the residual oxidants, such as ozone, hydroxide, or peroxide must make contact with the organisms in order for the disinfection action to proceed. Design of an ozone system as primary treatment should be based
on simple criteria, including:
(a) Ozone contact concentrations,
(b) Competing ozone demands
(c) Minimum contact concentration-time (CT) to meet the required inactivation requirements, in combination with US EPA recommendations.
Ozone has been observed to be capable of inactivating Cryptosporidium and there is significant interest in this aspect of its application [10]. Similar findings have been reported for the control of cyanobacterial toxins (microcystins) under various bloom conditions [11]. Available data indicate that a significant increase in ozone dose (at least 1.5 mg/L) and CT may be required as compared with past practices. Therefore, these needs in addition to continuous monitoring and ensuring a low total organic carbon
(TOC) in the flow should be considered in planning.
4.2. Zebra Mussel Abatement
Zebra mussel (Dreissena polymorpha), has arrived in the United States by attaching itself to ships in the infested waters of its natural habitat. Subsequently, it was imported over by the ships to cause infestation in US waters. The growth of zebra mussels on raw water intake pipes decreases the capacity of water transmission to potable-water-treatment plants, cooling towers of power plants, and hatcheries. To restore the full water flow capacity, plant operators have resorted to mechanical means for removing the mussel infestation on intake pipes.
Ozonation seems to be the most promising among the chemical alternatives tested for controlling zebra mussel growth. An 11 MGD side stream ozonation process was designed for a fish culture plant in New England [12]. It involved the pumping of high concentration ozone in water solution (15–25 mg/L) into the intake of the raw water pipe and blending it into the intake water to attain a final ozone concentration of 0.1–0.3 mg/L, which is sufficient to control the zebra mussel infestation.
4.3. Iron and Manganese Removal
The standard oxidation–reduction potential and reaction rate of ozone is such that it can readily oxidize iron and manganese in groundwater and in water with low organic content. Groundwater systems that have iron levels above 0.1 mg/L may have iron complaints if ozonation or chlorination is added. Excessive doses of ozone will lead to the formation of permanganate, which gives water a pinkish color. This soluble form of manganese (Mn) corresponds to a theoretical stoichiometry of 2.20 mg O3/mg Mn.
3.4. Color Removal
Because humic substances are the primary cause of color in natural waters, it is useful to review the reactions of ozone with humic and fulvic acids. According to different authors, ozone doses of 1–3 mg O3/mg C lead to almost complete color removal. The ozone dosages to be applied in order to reach treatment goals for color can be very high.
It is interesting to note that when the ozone dosage is sufficient, the organic structure is modified such that the final chlorine demand can decrease. Konsowa [13]. found that ozone is efficient in the removal of color from textile dyeing wastewater. The rate of dye oxidation was determined to be a function of dye concentration, ozone concentration, ozone-air flow rate and pH. The products produced by the break down of the dye are nontoxic and can be removed by biological treatment.
4.5. Control of Taste and Odor
The National Secondary Drinking Water Regulations recommend that the threshold odors number (TON) be 3 or less in finished water. It has been shown that ozone can be effective in treating water for taste and odor problems, especially when the water is relatively free from radical scavengers.
It has also been observed that ozone, in combination with other downstream treatment processes, especially granular activated carbon (GAC) filtration, can greatly increase taste and odor treatment efficiency and reliability. Again, the causes of taste and odor compounds, as well as the source water to be treated, need to be carefully considered prior to designing a treatment system. Analysis and possibly pilot-scale experimentation may be required to determine the optimum choice of ozone and downstream treatment.
4.6. Elimination of Organic Chemicals
Ozone or advanced ozonation processes can remove many synthetic organic chemicals (SOC). This removal leads to the chemical transformation of these molecules into toxic or nontoxic by-products. Such transformation can theoretically lead to complete oxidation into carbon dioxide (CO2); however, this is rarely the case in water treatment. Any observable reduction in total organic carbon (TOC) is due to either a small degree of CO2 formation (for example, decarboxylation of amino acids) or the formation and loss of volatile compounds through stripping.
4.7. Control of Algae
Ozone, like any other oxidant, such as chlorine or chlorine dioxide, has a lethal effect on some algae or limits its growth. Ozone is also capable of inactivating certain zooplankton,e.g., mobile organisms, Notholca caudata. Such organisms must first be inactivated before they are removed by flocculation and filtration.
4.8. Aid in Coagulation and Destabilization of Turbidity
It is important to understand that the coagulating effects of ozone go beyond any direct oxidative effects on organic macro-pollutants. For this reason, one must be wary of studies claiming improved removal of organic matter when the data are based solely on color removal or ultraviolet (UV) absorption. Also, when studying the removal of DBP such as trihalomethanes, one must be careful to incorporate controls permitting the separate evaluation of ozone’s direct effects. Finally, the coagulating effects of ozone may not be observed with all waters. Whenever considering the use of ozone as a coagulant aid, the preozonation effects should be critically evaluated in pilot studies incorporating the proper controls.
5.Disinfection of Wastewater by Ozone
As mentioned above, transfer of ozone into the wastewater is the first step in meeting the disinfection objective. Once transferred, the residual oxidants must make contact with the microorganisms in order for the disinfection action to proceed. Therefore, similar requirements and kinetic relationships used for chlorine disinfectants can also be used for ozone disinfection.
5.1. Wastewater Treatment Prior to Ozonation
The US EPA Water Engineering Research Laboratory and other researchers have evaluated several treatment plant effluents to determine the relationship between ozone dosage and total coliform reduction [14–18]. The most significant factor influencing the ozone dosage requirement to achieve a desired effluent total coliform concentration was the TCOD (total chemical oxygen demand) concentration of the effluent. For example, at five plants where the TCOD of the secondary effluent was less than 40 mg/L, a total coliform concentration of 1000 per 100 mL could be achieved with ozone dosages between 4 and 7 mg/L [18] .However, when Meckes et al. [18] evaluated a plant, which treated a significant amount of industrial waste (TCOD = 74 mg/L), a dosage greater than 12 mg/L was projected in order for the process to meet the 1000 total coliforms per 100 mL limit.
Based on the results of the above researchers, it appears that there is no technical basis for excluding the use of ozone following any treatment scheme (primary, secondary, tertiary, or advanced). However, depending on the type of wastewater treated and/or the effluent disinfection requirement, the wastewater-treatment scheme may be an important economical consideration. A summary of the issues to consider when selecting the wastewater-treatment scheme prior to ozone disinfection is presented below:
(a) Required Effluent Target
First: To meet the former US EPA standard of 200 fecal coliforms per 100 mL, tertiary treatment may not be necessary.
Second: To meet more stringent standards, such as 14 fecal coliforms per 100 mL, tertiary treatment should be considered.
Third: To meet a standard of 2.2 total coliforms per 100 mL, advanced treatment processes prior to the ozone disinfection process may be required.
(b) Influent Coliform Concentration
First: Coliform removal is a function of transferred ozone dosage; thus, the influent coliform concentration will affect the amount of ozone dosage required to meet specific effluent criteria.
Second: Treatment processes that reduce the influent coliform concentration (such as filtration) will decrease the ozone dosage required to achieve a specific effluent standard.
(c) Wastewater Quality Characteristics
First: The ozone demand of the wastewater significantly increases the ozone dosage requirements. A plant with a large industrial contribution may have a large ozone dosage requirement. Pilot testing to establish ozone dosage requirements in these plants is highly recommended.
Second: Incomplete nitrification and a high concentration of nitrite-nitrogen will significantly
increase the ozone demand and thus the ozone dosage requirement. The nitrite-nitrogen concentration preferably should be less than 0.15 mg/L to optimize disinfection performance.

By
Ahmed Ahmed Elserwy
Water & Environmental Consultant
Ain Shames University, Faculty of Science.
References
[1] C. Gottschalk, J. A. Libra, and A. Saupe, Ozonation of Water and Waste Water: A Practical
Guide to Understanding Ozone and its Application, Wiley-VCH, New York, NY, 2000.
[2] US EPA, Small System Treatment Technologies for Surface Water and Total Coliform Rules,US EPA Office of Ground Water and Drinking Water, Washington, DC, 1998.
[3] US EPA, Design Manual—Municipal Wastewater Disinfection, US Environmental Protection Agency, EPA/625 1-86-021, Water Engineering Research Laboratory, Cincinnati,OH, 1986.
[4] G. W. Miller, et al., An Assessment of Ozone and Chlorine Dioxide Technologies for Treatment of Municipal Water Supplies, EPA-600/2-78-147, US Environmental Protection Agency, Cincinnati, OH, 1978.
[5] NDWC Tech brief: Ozone, Newsletter On Tap, National Drinking Water Clearinghouse (NDWC), No. 12, December, (1999).
[6] X. Paraskeva, Y. Panagiota, Z. Graham, J. D. Nigel, Water Environment Regulation Watch, November/December, (2002).
[7] M. R. Collins, Small Systems Water Treatment Technologies: State-of-the-Art Workshop ,American Water Works Association, Denver, CO, 1998.
[8] Tri-State (2003), Ozonation Systems, Energy Technologies, Generation and Transmission Association, http://tristate.apogee.net/et/ewtwozs.asp, November, (2003).
[9] S. Farooq, et al., Criteria of Design of Ozone Disinfection Plants, Forum on Ozone Disinfection, International Ozone Institute, 1976.
[10] SNWA (2003), Water Quality and Water Treatment: Ozonation, Southern Nevada Water Authority, http://www.snwa.com/html/wq_treatment_ozonation.html, November.
[11] S. J. Hoeger, D. R. Dietrich, and B. C. Hitzfeld Environmental Health Perspectives, 110,November (2002).
[12] Bollyky Associates, Inc. Zebra Mussel Control by Ozone Treatment, http://www.bai-ozone.com/bai 11.htm, November, (2003).
[13] A. H. Konsowa, Desalination, 158, 233–240, (2003).
[14] E. L. Stover, et al., High Level Ozone Disinfection of Municipal Wastewater Effluents, US EPA Grant No. R8O4946, 1980.
[15] P. W. Given and D. W. Smith, In: Municipal Wastewater Disinfection, Proceedings of Second National Symposium, Orlando, Florida, EPA-600/9-83-009, US Environmental Protection Agency, Cincinnati, OH, 1983.
[16] H. B. Gan, et al., Forum on Ozone Disinfection, International Ozone Institute, 1976.
[17] A. D. Venosa, et al., Environment International, 4, 299–311, (1980).
[18] M. C. Meckes, et al., J. Water Pollution Control Fed., 55, 1158–1162 (1983).